Colony PCRs
Protocol: PCR
PCR is a reaction that makes it possible (and relatively easy) to amplify
a certain region of DNA. The first step is the selection of that region
(and the design of the relevant primers). Primer design can be done by hand, or by
using our Primer Design Helper. Once
done, order the primers (in our case, we ordered from them IDT).
When you've received the primers, prepare them and make sure you've got your PCR kit (we used the "Phusion® High-Fidelity DNA Polymerase"). Start preparing your master mix, the composition for one tube is:
1X Mastermix 20μl reaction, add in this order
| Reagent | Volume [μl] |
|---|---|
| Water | Complete to total volume of 20μl |
| HF-Buffer (5x) | 4 |
| DMSO (optional) | 0.6 |
| dNTPs | 0.4 |
| Forward primer (50μM) | 0.2 |
| Reverse primer (50μM) | 0.2 |
| Template (10ng/μl) | 0.5 |
| Phusion HF polymerase | 0.2 |
Prepare one or two extra tubes-worth of reagent (you'll use some liquid on the walls of your tips).
Once you've finished, you should run the resulting products on a gel to check if everything went as planned.
Tips
- Thaw the HF-Buffer, DMSO and dNTPs before making the mastermix.
- Avoid taking the Phusion-HF polymerase out of the freezer (only take it out briefly when you need to add it).
- If the reactions have different primers and/or template, add the polymerase right after the dNTPs, split the mastermix and add the rest.
- Don't forget positive and negative controls
- Primers should have similar Tms (less than 5°C).
- Primer Tm calculation is a less exact science than it should be (just test several tools and compare their results). If you're not sure what the correct Tm is, consider using a gradient PCR.
- Avoid primers with strong secondary structures.
- PCR can introduce mutations. Don't forget to sequence your final product (this could be your final plasmid): you really don't want to lose a few weeks because of a "corrupt" plasmid.
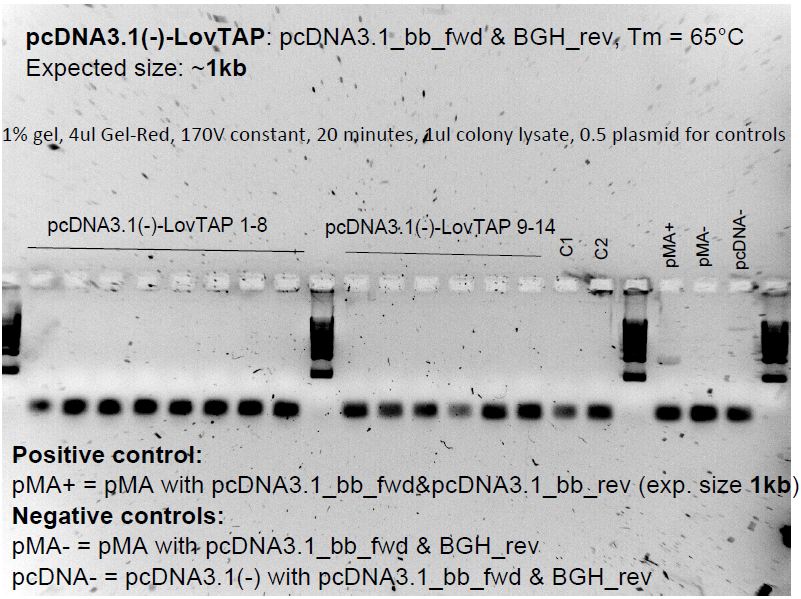
Colonies were present on pcDNA3.1(-)-LovTAP-BB plate from last night's transformation. They were picked up and underwent a colony PCR, then were run on a gel. Colonies on the pGL-SEAP plates were picked up and PCR'd too. The gel was run on the following day.
Gel electrophoresis of pcDNA3.1(-)-LovTAP-BB colony PCR
Protocol: Gel Electrophoresis
Agarose concentration depends on the size of the DNA to be run. We will mostly use 1%.
VOL is the desired volume of gel in ml:
CH Lab
- Add 0.01*VOL g of agarose to a clean glass bottle.
- Pour VOL/50 ml of 50xTAE in a graduated cylinder. Fill up to VOL ml with di water.
- Add the resulting VOL ml of 1xTAE to the glass bottle with agarose.
- Microwave, at 7, the bottle (loose cap!) until it boils.
- Carefully remove bottle (can be super heated!) and check for the total absence of particles. Microwave again if needed.
- Prepare a gel box, with comb, and fill it up with the agarose solution (maybe not the whole solution is needed).
- Add 0.05 µl per ml of gel in the box of Red Gel (it's in the iGEM drawer) and stirr until disolved.
- Wait until cold and solidified.
- Carefully remove comb.
- Place the box in the electrophoresis chamber.
- Fill up the electrophresis chamber with 1x TAE buffer.
- Add blue dye to the DNA samples (6x loading buffer, that is 10 µl in 50 µl of DNA solution).
- Inject 30 µl of ladder marker in the first well (that's 1 µg of DNA).
- Inject 60 µl of each DNA solution in the other wells.
- Set voltage to 70-90 V and run for 30-40 min, or until the dye reaches the last 25% of the gel length (DNA travels from - to +).
- Place the gel under the camera, cover, turn UV on and take photos!
Preparing the ladder:
- get 1kb ladder DNA from the freezer (500 µg/ml).
- for 30 charges, 30 µl per charge, we need 900 µl:
- 60 µl of 1kb ladder DNA
- 150 µl of dye (6x loading buffer)
- 690 µl of water
BM Lab
In this lab the gels are slightly different. The total volumes for the small, the medium and the large gel are respectively 60ml, 80ml and 90ml. As we use 0.5x TAE buffer instead of 1x, we can use higher voltages (170V seems to work fine). The gel should run 20-40 minutes, not more. As the gel is thinner, load less DNA (up to ~10ul).
This was an attempt to put our BioBrick (with its BioBrick ends) into a BioBrick-compatible plasmid to be able to test it in mammalian cells. It didn't work, and we settled with the original LovTAP in pcDNA3.1(+), which is also a very good testing subject.
Biobricking PCR
Protocol: PCR
PCR is a reaction that makes it possible (and relatively easy) to amplify
a certain region of DNA. The first step is the selection of that region
(and the design of the relevant primers). Primer design can be done by hand, or by
using our Primer Design Helper. Once
done, order the primers (in our case, we ordered from them IDT).
When you've received the primers, prepare them and make sure you've got your PCR kit (we used the "Phusion® High-Fidelity DNA Polymerase"). Start preparing your master mix, the composition for one tube is:
1X Mastermix 20μl reaction, add in this order
| Reagent | Volume [μl] |
|---|---|
| Water | Complete to total volume of 20μl |
| HF-Buffer (5x) | 4 |
| DMSO (optional) | 0.6 |
| dNTPs | 0.4 |
| Forward primer (50μM) | 0.2 |
| Reverse primer (50μM) | 0.2 |
| Template (10ng/μl) | 0.5 |
| Phusion HF polymerase | 0.2 |
Prepare one or two extra tubes-worth of reagent (you'll use some liquid on the walls of your tips).
Once you've finished, you should run the resulting products on a gel to check if everything went as planned.
Tips
- Thaw the HF-Buffer, DMSO and dNTPs before making the mastermix.
- Avoid taking the Phusion-HF polymerase out of the freezer (only take it out briefly when you need to add it).
- If the reactions have different primers and/or template, add the polymerase right after the dNTPs, split the mastermix and add the rest.
- Don't forget positive and negative controls
- Primers should have similar Tms (less than 5°C).
- Primer Tm calculation is a less exact science than it should be (just test several tools and compare their results). If you're not sure what the correct Tm is, consider using a gradient PCR.
- Avoid primers with strong secondary structures.
- PCR can introduce mutations. Don't forget to sequence your final product (this could be your final plasmid): you really don't want to lose a few weeks because of a "corrupt" plasmid.
We ran a PCR that adds BioBrick tails to the readout for the LovTAP experiment (dsRed).
Gel electrophoresis of the RO BioBrick
Protocol: Gel Electrophoresis
Agarose concentration depends on the size of the DNA to be run. We will mostly use 1%.
VOL is the desired volume of gel in ml:
CH Lab
- Add 0.01*VOL g of agarose to a clean glass bottle.
- Pour VOL/50 ml of 50xTAE in a graduated cylinder. Fill up to VOL ml with di water.
- Add the resulting VOL ml of 1xTAE to the glass bottle with agarose.
- Microwave, at 7, the bottle (loose cap!) until it boils.
- Carefully remove bottle (can be super heated!) and check for the total absence of particles. Microwave again if needed.
- Prepare a gel box, with comb, and fill it up with the agarose solution (maybe not the whole solution is needed).
- Add 0.05 µl per ml of gel in the box of Red Gel (it's in the iGEM drawer) and stirr until disolved.
- Wait until cold and solidified.
- Carefully remove comb.
- Place the box in the electrophoresis chamber.
- Fill up the electrophresis chamber with 1x TAE buffer.
- Add blue dye to the DNA samples (6x loading buffer, that is 10 µl in 50 µl of DNA solution).
- Inject 30 µl of ladder marker in the first well (that's 1 µg of DNA).
- Inject 60 µl of each DNA solution in the other wells.
- Set voltage to 70-90 V and run for 30-40 min, or until the dye reaches the last 25% of the gel length (DNA travels from - to +).
- Place the gel under the camera, cover, turn UV on and take photos!
Preparing the ladder:
- get 1kb ladder DNA from the freezer (500 µg/ml).
- for 30 charges, 30 µl per charge, we need 900 µl:
- 60 µl of 1kb ladder DNA
- 150 µl of dye (6x loading buffer)
- 690 µl of water
BM Lab
In this lab the gels are slightly different. The total volumes for the small, the medium and the large gel are respectively 60ml, 80ml and 90ml. As we use 0.5x TAE buffer instead of 1x, we can use higher voltages (170V seems to work fine). The gel should run 20-40 minutes, not more. As the gel is thinner, load less DNA (up to ~10ul).
The PCR apparently did not work, we need to try another gradient.
Restriction digest of pCEP4, melanopsin, SEAP and TNFR
Protocol: Restriction site digestion
- Look for the best pair of restriction sites, ideally with similar digestion temperatures and times.
- NEBcutter for finding cutting enzymes.
- Double Digest Finder for the parameters.
- Calculate the amounts required of:
- DNA
- Buffer (usually from 10x to 1x)
- BSA, if needed (usually from 100x to 1x)
- Enzymes (depends on the amount of DNA)
- Water
- Get the recommended buffer (and BSA if needed) from the freezer and let defreeze.
- Mix all the ingredients, except DNA, in a tube.
- Note: Enzymes should stay no longer than a couple of minutes out of the freezer. Don't touch the bottom of the tubes! Don't vortex!
- Distribute the mix in as many tubes as DNA samples and add the DNA.
- Keep in the Thermomixer at the recommended temperature.
Sowmya's recommended amounts (50 µl total solution):
- 5 µl of 10x buffer
- 0.5 µl of 100x BSA
- 1 µl of each enzyme
- 5 µl of DNA
- 37.5 (up to 50 µl) of water.
Protocol based on what was done on July the 4th.
- pCEP4-HA (and also pCEP4, we have two different versions of this plasmid, one of them has a tag) were digested with KpnI and BglII for insertion of the readout.
- The melanopsin PCR fragment in pHY42 was digested with EcoRI and SpeI for biobricking.
- The SEAP PCR fragment was digested with EcoRI and SpeI for biobricking.
- The TNFR PCR fragment was digested with EcoRI and SpeI for biobricking.
Gel electrophoresis of the digested backbone
Protocol: Gel Electrophoresis
Agarose concentration depends on the size of the DNA to be run. We will mostly use 1%.
VOL is the desired volume of gel in ml:
CH Lab
- Add 0.01*VOL g of agarose to a clean glass bottle.
- Pour VOL/50 ml of 50xTAE in a graduated cylinder. Fill up to VOL ml with di water.
- Add the resulting VOL ml of 1xTAE to the glass bottle with agarose.
- Microwave, at 7, the bottle (loose cap!) until it boils.
- Carefully remove bottle (can be super heated!) and check for the total absence of particles. Microwave again if needed.
- Prepare a gel box, with comb, and fill it up with the agarose solution (maybe not the whole solution is needed).
- Add 0.05 µl per ml of gel in the box of Red Gel (it's in the iGEM drawer) and stirr until disolved.
- Wait until cold and solidified.
- Carefully remove comb.
- Place the box in the electrophoresis chamber.
- Fill up the electrophresis chamber with 1x TAE buffer.
- Add blue dye to the DNA samples (6x loading buffer, that is 10 µl in 50 µl of DNA solution).
- Inject 30 µl of ladder marker in the first well (that's 1 µg of DNA).
- Inject 60 µl of each DNA solution in the other wells.
- Set voltage to 70-90 V and run for 30-40 min, or until the dye reaches the last 25% of the gel length (DNA travels from - to +).
- Place the gel under the camera, cover, turn UV on and take photos!
Preparing the ladder:
- get 1kb ladder DNA from the freezer (500 µg/ml).
- for 30 charges, 30 µl per charge, we need 900 µl:
- 60 µl of 1kb ladder DNA
- 150 µl of dye (6x loading buffer)
- 690 µl of water
BM Lab
In this lab the gels are slightly different. The total volumes for the small, the medium and the large gel are respectively 60ml, 80ml and 90ml. As we use 0.5x TAE buffer instead of 1x, we can use higher voltages (170V seems to work fine). The gel should run 20-40 minutes, not more. As the gel is thinner, load less DNA (up to ~10ul).
The digested pCEP4-HA was run on a gel.
Gel extraction
Protocol: Gel Extraction
A gel extraction is used to select a fragment of DNA of a specific length
out of a solution composed of different fragments (ideally the difference
in length between the wanted fragment and the closest-sized fragment should
be more than 200bp). These fragments are often obtained after a
digestion.
The yeild for this procedure is typically very poor so a large amount of starting material, digested DNA in this case is required. We typically used 4 micrograms. The digestion products are loaded on a gel. Lanes on both sides of the one to be extracted should be empty to make cutting easier and avoid contamination with other fragments.
The gel should be run long enough for the bands to be spread out. This is particularly important if the fragment of interest is around the same length as other expected digestion products. UV light is necesary to observe the bands on the gel but exposure time should be minimized to avoid DNA damage. The fragment of interest is then excise and put in an Eppendorf (consider using a 2ml one).
To extract the DNA from the agarose we used Macherey-Nagel's "Nucleospin® Gel and PCR clean-up" kit. Its manual can be found here: Gel and PCR clean-up Manual
Tips
- Cut away as much Agar as possible without slicing into the DNA. Excess agar will require more solvent to dissolve and will result in a poorer yeild upon elution.
- Minimize the DNA's exposure to the UV-light. UV will damage DNA and have negative effects on any subsequent reactions (for example, ligations can be 10'000x less effective when DNA has been exposed to too much UV light [1]
The pCEP4 and pCEP4-HA backbone were excised from the gel you can see above and purified using a Macherey-Nagel PCR purification kit.
 "
"
